Theoretical Approach
Scientific Paper
This analysis was based on the scientific paper “The Role of Osteocytes in Targeted Bone Remodeling: A Mathematical Model” by Jason M. Graham, Bruce P. Ayati, Sarah A. Holstein, James. A. Martin [1], available in http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0063884.
Glossary
Osteocytes: Most abundant bone cells, inserted in the bone [2]. They product type I collagen and other substances to form the extracellular matrix [3].
Pre-Osteoblasts: Mesenchymal cells, located in the inner layer, that develop into osteoblasts [4]. They secrete osteoid, an organic non-mineralized part of the bone matrix that is produced before the maturation of the bone [5].
Osteoblasts: Large bone cells, found in the surface of bone, responsible for the synthesis and mineralization of bone formation and bone remodeling. They can differentiate into osteocytes. [3] [6].
Osteoclasts: Large multinucleated bone cell, descendent from stem cells, responsible for the bone resorption. They occupy small depressions on the surface of the bone [3] [7].
Bone Remodeling: Regulated process of resorption by osteoclasts followed by replacement of new bone by osteoblasts [8] [9].
Bone Resorption: Stage of the bone remodeling process where the old bone is broken down and absorbed [10].
Sclerostin: Formed primarily by osteocytes, it inhibits the bone formation and stimulates apoptosis of osteoblasts [11].
Receptor Activator of Nuclear Factor κ-B Ligand (RANKL): it binds to the Receptor Activator of Nuclear Factor κ-B (RANK) on osteoclast precursors and leads to the maturation of osteoclasts. It is secreted by osteoblasts [12].
Osteoprotegerin (OPG): receptor secreted by osteoblasts and sequesters RANKL, inhibiting osteoclast [12]. It is produced by differentiated osteoblastic cells [1].
Wnt/β-catenin pathway: A signaling pathway that stimulates pre-osteoblasts replication and osteoblastogenesis and inhibits osteocytes and osteoblasts apoptosis [13].
Content Review
Bone remodeling, a process that undergoes continuously through life, involves the removal of old bone by osteoclasts followed by the formation of bone matrix through the osteoblasts [9] [14]. [15]. Thus, it can be defined as an active process that balances the bone resorption and the bone formation [12].
Bone resorption is assured by active osteoclasts, who are, in normal conditions, stimulated by RANKL. On the other hand, bone formation is achieved by inhibiting osteoclasts, through OPG, and stimulating osteoblasts [12].
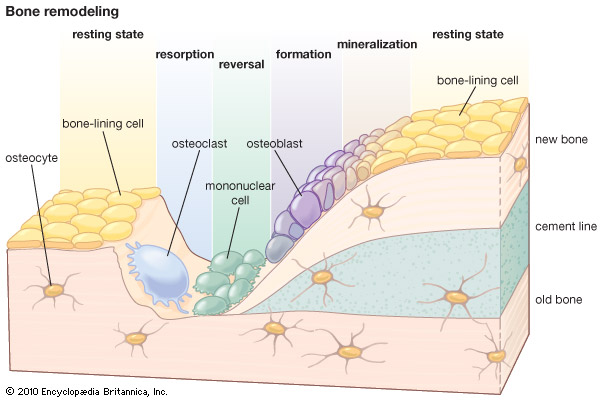
Figure 1: Bone remodeling. Taken from. [15].
Until recently, not much attention was given to the role of osteocytes in bone remodeling. However, as discussed in [1], there is increasing evidence that it has a fundamental role secreting sclerostin and being also a source of RANKL [1].
It has been suggested that sclerostin is a major mediator in bone remodeling, through two opposite mechanisms: high sclerostin levels can lead to an antagonization of Wnt/β-catenin signaling pathway and direct to bone resorption; low sclerostin levels can activate Wnt/β-catenin signaling pathway and lead to bone formation [16].
Computational Simulation
Algorithm Analysis
The following analysis is based on what is referred and reported in [1]. The symbols used in the equations, their definition and their values are in Table 1, which were transcribed from [1]. We will focus our analysis, and following simulation, in the baseline case presented in [1].
Table 1: The parameters used in the equations and their definition and values. Transcribed from [1]
| Parameter | Definition | Value |
|---|---|---|
| α1 | Osteoblast embedding rate | 0.5 per day |
| α2 | Differentiation rate of pre-osteoblast precursors | 0.1 per day |
| α3 | Pre-osteoblasts proliferation rate | 0.1 per day |
| β1 | Differentiation rate of pre-osteoblasts | 0.1 per day |
| δ | Apoptosis of pre-osteoblasts | 0.1 per day |
| β2 | Osteoblast apoptosis | 0.1 per day |
| α4 | Differentiation rate of osteoclasts precursors | 0.1 per day |
| KS | Critical value of osteocyte population | 200 cells |
| k1 | Bone resorption rate | 0.7 % volume per day |
| k2 | Bone formation rate | 0.015445 % volume per day |
| g31 | Effectiveness of osteoblast autocrine signaling | 1 (dimensionless) |
| g21 | Effectiveness of osteocyte paracrine signaling of osteoblasts | 2 (dimensionless) |
| g22 | Effectiveness of sclerostin regulation of osteoblastogenesis | 1 (dimensionless) |
| g32 | Effectiveness of pre-osteoblasts autocrine signaling | 1 (dimensionless) |
| g41 | Effectiveness of osteocyte paracrine signaling of osteoclasts | 1 (dimensionless) |
| g42 | Effectiveness of sclerostin regulation of osteoclastogenesis | 1 (dimensionless) |
| g43 | Effectiveness of osteoblast paracrine signaling of osteoclasts | -1 (dimensionless) |
| g44 | Effectiveness of sclerostin regulation of osteoclastogenesis | 1 (dimensionless) |
| f12 | Effectiveness of pre-osteoblast paracrine signaling of osteoblasts | 1 (dimensionless) |
| f14 | Effectiveness of osteoclasts paracrine signaling of osteoblasts | 1 (dimensionless) |
| f23 | Effectiveness of osteoblasts autocrine signaling for apoptosis | 1 (dimensionless) |
| f34 | Effectiveness of osteoclasts autocrine signaling for apoptosis | 1 (dimensionless) |
Jason M. Graham, Bruce P. Ayati, Sarah A. Holstein and James A. Martin established a cell population model for osteocyte-induced targeted bone remodeling. This resides in an osteocyte, a pre-osteoblast, an osteoblast and an osteoclast cell population, the interactions of these cells and relation of the autocrine and paracrine signaling among these cells [1].
dS/dt = α1Bg31 (1- S/KS)+ (1)
dP/dt = α2Sg21 (1- S/KS)+g22 + α3Pg32 (1- S/KS)+ - β1Pf12Cf14 - δP (2)
dB/dt = β1Pf12Cf14 - β2Bf23 - α1Bg31 (1- S/KS)+ (3)
dC/dt = α4Sg41Pg42 (ε + B)g43 (1- S/KS)+g44 - β3Cf34 (4)
dz/dt = -k1C + k2B (5)
Equation 1, Equation 2, Equation 3 and Equation 4 describe the dynamics, respectively, of the osteocyte, pre-osteoblast, osteoblast and osteoclast cell population at a time t. Equation 5 defines the bone mass at a given time t [1].
The initial conditions presented in the scientific paper for the mathematical model, and used in the simulation, are: S(0) = KS – ρ, where ρ>0 is a constant that denotes the osteocytes apoptosis, P(0)=B(0)=C(0) and z(0)=200 [1].
Results Interpretation
The simulations presented in [1] were replicated for the baseline case and the graphics obtained are identical to those displayed in the scientific paper.
The graphics show the simulation results for bone cell populations and the dynamics of bone volume during a complete bone remodeling that occurs over a period of hundred days, in which the bone resorption is equal to the bone formation [1].
Graphic 1 shows the dynamics of all cell populations. The level of osteocytes is initially low, representing the cells’ apoptosis and the subsequent decreasing in local sclerostin levels, which leads to the proliferation and differentiation of pre-osteoblasts. The differentiation of these pre-osteoblasts leads to the increasing of the number of osteoblasts. This goes along with the differentiation of osteoclast precursors to mature osteoclasts. Once the bone remodeling finishes, all the cell population to their initial values, except for osteocytes which remain in their steady state value [1].
Graphic 2 represents the dynamics of bone volume, illustrating the resorption and formation of bone. As it can be seen, and because of the aforementioned, the old bone resorption is equal to the new bone formation, at the end of the process [1].
Online Experiment
This animation shows the process of bone remodeling. It was created by Susan Ott and can be found in [18].
References
[INTRO IMAGE] “Haversian System,” http://medcell.med.yale.edu/, [Online]. Available: http://medcell.med.yale.edu/image_gallery/load_image.php?lab=bone&image=haversian_system. [Accessed 27 August 2016].
[1] Jason M. Graham, Bruce P. Ayati, Sarah A. Holstein, James A. Martin, “The Role of Osteocytes in Targeted Bone Remodeling: A Mathematical Model,” PloS one, vol. 8, nº 5, p. e63884, 2013.
[2] Jenneke Klein-Nulend, Peter J. Nijweide, Elisabeth H. Burger, “Osteocyte and bone structure,” Current Osteoporosis Reports, vol. 1, nº 1, pp. 5-10, 2003.
[3] “Bone Remodeling,” UW Courses Web Server, [Online]. Available: https://courses.washington.edu/conj/bess/bone/bone2.html. [Accessed 27 August 2016].
[4] “osteoprogenitor cell,” Farlex, Inc, [Online]. Available: http://medical-dictionary.thefreedictionary.com/preosteoblast. [Accessed 27 August 2016].
[5] “Endochondral Preosteoblasts (CEBs),” LifeMap Sciences, Inc. , [Online]. Available: http://discovery.lifemapsc.com/in-vivo-development/bone/caudal-facial-endochondral-bones/endochondral-preosteoblasts. [Accessed 27 August 2016].
[6] The Editors of Encyclopædia Britannica, “Osteoblast,” Encyclopædia Britannica, Inc., [Online]. Available: https://www.britannica.com/science/osteoblast. [Accessed 27 August 2016].
[7] The Editors of Encyclopædia Britannica, “Osteoclast,” Encyclopædia Britannica, Inc., [Online]. Available: https://www.britannica.com/science/osteoclast. [Accessed 27 August 2016].
[8] Erik Fink Eriksen, “Cellular mechanisms of bone remodeling,” Reviews in Endocrine and Metabolic Disorders, vol. 11, nº 4, pp. 219-227, 2010.
[9] “Bone Remodeling,” Boundless.com, [Online]. Available: https://www.boundless.com/physiology/textbooks/boundless-anatomy-and-physiology-textbook/skeletal-system-6/bone-formation-72/bone-remodeling-441-8963/. [Accessed 27 August 2016].
[10] Osteoporosis-health, “Bone Resorption Definition,” Veritashealth.com, [Online]. Available: http://www.osteoporosis-health.com/glossary/bone-resorption. [Accessed 27 August 2016].
[11] “Sclerostin (SOST),” UW Courses Web Server, [Online]. Available: https://courses.washington.edu/bonephys/physsost.html. [Accessed 27 August 2016].
[12] Daniel Hatch, “Bone Signaling & RANKL,” orthobullets, 20 February 2016. [Online]. Available: http://www.orthobullets.com/basic-science/9010/bone-signaling-and-rankl. [Accessed 28 August 2016].
[13] Venkatesh Krishnan, Henry U. Bryant, Ormond A. MacDougald, “Regulation of bone mass by Wnt signaling,” The Journal of clinical investigation , vol. 116, nº 5, pp. 1202-1209, 2006.
[14] D. J. Hadjidakis, I.I. Androulakis, “Bone remodeling.,” Annals of the New York Academy of Sciences, vol. 1092, nº 1, pp. 385-396, 2006.
[15] The Editors of Encyclopædia Britannica, “Bone remodeling,” Encyclopædia Britannica, Inc., [Online]. Available: https://www.britannica.com/science/bone-remodeling. [Accessed 26 August 2016].
[16] R. Sapir-Koren, G. Livshits, “Osteocyte control of bone remodeling: is sclerostin a key molecular coordinator of the balanced bone resorption-formation cycles?,” Osteoporosis International, vol. 25, nº 12, pp. 2685-2700, 2014.
[17] Amgen, “Bone Remodeling and Modeling,” YouTube, 01 January 2012. [Online]. Available: https://www.youtube.com/watch?v=0dV1Bwe2v6c. [Accessed 26 August 2016].
[18] Susan Ott, “Bone remodeling,” UW Courses Web Server, 2008. [Online]. Available: https://courses.washington.edu/bonephys/physremod.html. [Accessed 29 August 2016].